Based on this structure, they simulated how Atg44 binds to lipid membranes and predicted that the monomers, dimers, and tetramers bind to lipid membranes using their hydrophobic face. To further investigate the cleavage mechanism, they analyzed the crystal structure of the Atg44 monomer and found that one side is hydrophilic, and the other side is hydrophobic. It was found that the factor has the ability to bind and cleave lipid membranes. When purified Atg44 was added to artificial lipid nanotubes that imitated the mitochondrial lipid membrane, the lipid nanotubes were cleaved upon binding to the lipid membrane over time. Due to this, they further analyzed its molecular function. They also confirmed that the factor serves as the mitochondrial fission factor when expressed in human cultured cells.īased on these results, this factor was suggested to have a function in initiating mitochondrial fission during mitophagy. Furthermore, in the deficient strain, another mitophagy protein marker (Atg32), previously reported by the research group, accumulated around the protrusions. In normal mitophagy, the isolating membrane extends to envelop the protrusions and engulf a part of the mitochondria when autophagosomes are formed however, in the deficient strain, this process halted leaving the extended autophagosome bound with the mitochondria. In yeast strains lacking Atg44, mitochondria were found to be swollen and enlarged.įluorescence and electron microscopy revealed that, when mitophagy was induced, protrusions similar to those observed during mitophagy accumulated in the deficient strain. They confirmed that the factor is localized at the mitochondrial intermembrane space. This factor, a small protein composed of 73 amino acids, is also present in budding yeast. 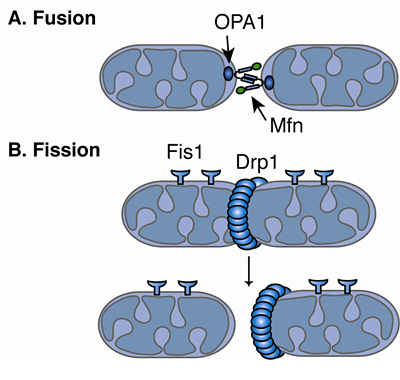
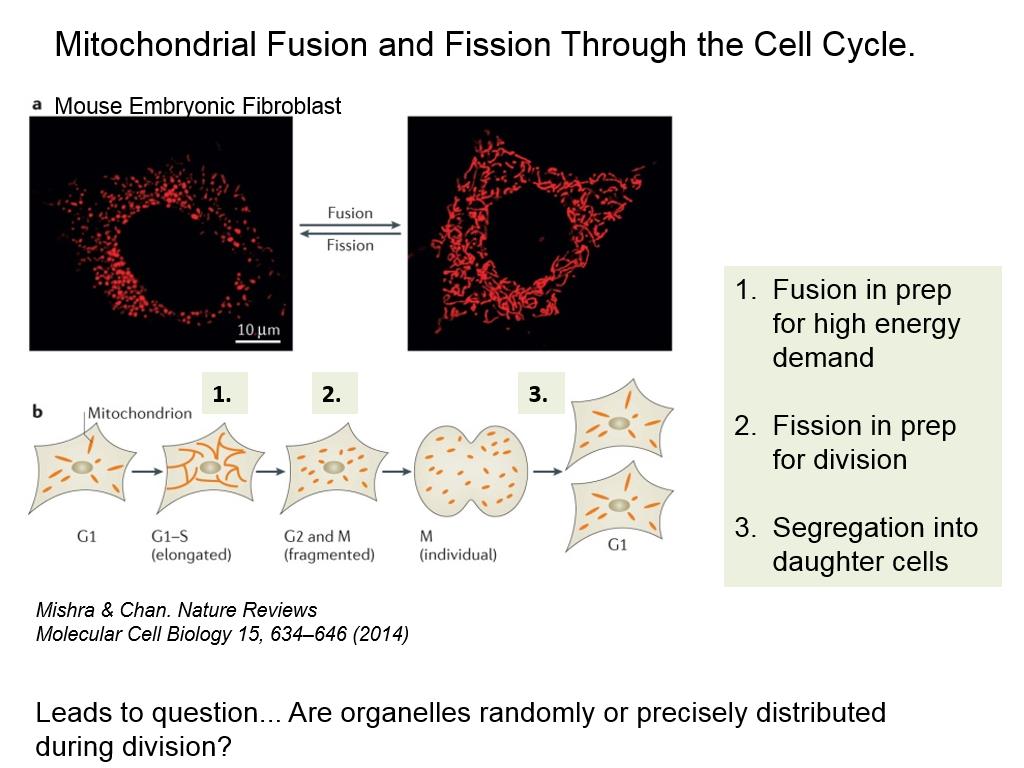
By establishing an experimental system in fission yeast and utilizing a gene disruption library, they discovered the Atg44 protein, an essential factor for mitophagy, and named it Mitofissin. In the recent study, the researchers used fission yeast instead of budding yeast, which is more common in autophagy and mitophagy research. The group revealed that mitophagy wraps only a part of the mitochondria and forms autophagosomes by a mechanism different from fission. In 2016, the research group reported that mitophagy does not require a dynamin-like protein that is essential for mitochondrial fission. Mitophagy is a phenomenon where autophagy selectively degrades mitochondria, and mitochondria of various sizes within cells are thought to maintain their quality through mitophagy, fission, and fusion. Mitofissin-mediated mitochondrial fission required for mitophagy. The results were published in the June 15th issue of the international academic journal Molecular Cell. These advancements are expected to clarify the molecular mechanism of mitophagy and develop novel treatments for mitochondrial diseases. They established an experimental system in fission yeast and found that Mitofissin cleaves the mitochondrial membrane during mitophagy and splits the membrane into a size that fits into the autophagosome. The group led by Associate Professor Tomoyuki Fukuda, Specially Appointed Lecturer Kentaro Furukawa, Professor Tomotake Kanki of the Graduate School of Medical and Dental Sciences at Niigata University, Researcher Tatsuro Maruyama of the Institute for Microbial Chemistry, and Professor Nobuo Noda of the Institute for Genetic Medicine at Hokkaido University announced the discovery of a novel mitochondrial fission factor, "Mitofissin" (mitochondrial fission protein), in collaboration with the National Institute for Physiological Sciences, RIKEN, and the University of Michigan in the United States.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
